Institute of Chemistry, Academia Sinica – Research
Directions
The research thrusts of the Institute are currently grouped along three major directions: materials chemistry, chemical synthesis and catalysis, and chemical biology. The current topics of materials sector include organic electroluminescent materials and devices, organic field-effect transistor materials and devices, photovoltaic materials and devices. The chemical catalysis and synthesis sector is focusing on the development of new synthetic methodology, drug discovery, carbohydrate chemistry, and the development of new catalytic systems for the generation of renewal energies and green fuels. The chemical biology program has made important advances in the delineation of bio-macromolecular structures and the development of new analytical platforms for disease detection and diagnosis.
Materials Chemistry: Organic Electronic and Optoelectronic Materialsy
Applications of organic optoelectronic materials and molecular engineering of nanomaterials are two major research directions under the materials division. Noticeable results include developments of blue fluorescent molecular materials for high performance organic light-emitting diodes, rational design of field-effective organic memory devices based on pentacene and gold nanoparticles, the very first stable organic thin film transistor based on single crystal of hexacene, rare single-walled metal–organic nanotube (MONT) with a large exterior wall diameter, the applications of metal-organic framework as optoelectronic materials, and a number of high performance materials for efficient solar energy harvesting devices such as dye-sensitized solar cells, perovskite solar cells, or organic photovoltaics. Researchers in this sector also develop stimuli-responsive materials, core-shell nanomaterials, and biomaterials. A recent report shows that a cell membrane–mimicking conducting polymer is capable to integrate biochemical and electrical stimulation to promote neural cellular behavior with great enhancement of neurite outgrowth on this conducting polymer.
Chemical Catalysis and Synthesis: Green Catalysis and Synthetic Methodology
In response to the increasingly demands of sustainable fuel and green synthetic technology, researchers in the organic synthesis and chemical catalysis divisions have strived to advance the development of cutting-edge technology for chemical transformations. The synthetic chemistry division of this sector focuses on the advances of synthetic methodology and drug discovery. The research topics under catalysis division is reconciling to catalysis relating to renewable energy. Major research directions in this sector include: (1) synthetic methodology: silyl ethers for hydroxy- directed nucleophilic acyl alkylation, microwave-assisted carbohydrate synthesis, smart fluorescent probes for bioorthogonal sugar labeling; (2) coordination chemistry: approaching unconventional catalysis via amino-NHC and carbodicarbene, unconventional porphyrin complexes for small molecule activations, engineering cytochrome P450 BM3 and alkane hydroxylase (AlkB) for alkane oxidations; (3) renewable energy catalysis: catalytic hydrogen evolution and mechanistic studies, encapsulated tricopper cluster for methane to methanol conversion, and novel catalysts for valorization of lignocellulosic biomass feedstocks.
Chemical Biology: New Material and Method towards Sustainable Health
Chemical biology division focus on the development of new material and methodology to explore the structure and function of macromolecules associated with cellular function or human diseases. The research activities are directed to unravel the underlying pathological mechanism and to derive new diagnostic and therapeutic strategies. Research topics in this division cover (1) development of smart biomaterials based on novel molecular principles; (2) chemical probe and advanced techniques in bio-imaging and structural biology; (3) drug discovery in cancer, infectious and neurodegenerative diseases; (4) development of structural biology techniques for infectious diseases, and (5) development of advanced proteomics strategies for biomarker discovery. The major achievements from the chemical biology group include the establishment of multiplexed quantitative strategy for membrane proteomics and post-translational modification for delineating disease mechanism and mining therapeutic targets discovery of amyloid fibrils induced from the TDP-43 in the Amyotrophic Lateral Sclerosis (ALS), and development of a photocontrollable probe to induce TDP-43 aggregates in live cells, mapping of the RNA exit channel on transcribing RNA polymerase II by FRET analysis, development of nano velcro chip to capture circulating tumor cells for liquid biopsy, construction of a near-infrared- activatable enzyme platform using an up-conversion nanoparticle to remotely trigger intracellular signal transduction.
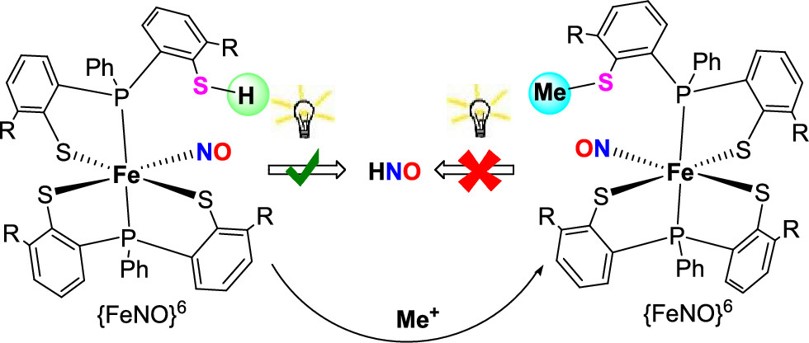
以垂掛硫醇之單核鐵觸媒 {FeNO}6 做光催化 NO 與 HNO 產生
Photoinduced NO and HNO Production from Mononuclear {FeNO}6 Complex Bearing a Pendant ThiolJ. Am. Chem. Soc. 2020, 142, 8649−8661. Chuan-Kuei Chiang, Kai-Ti Chu, Chia-Chin Lin, Shi-Rou Xie, Yu-Chiao Liu, Serhiy Demeshko, Gene-Hsiang Lee, Franc Meyer, Ming-Li Tsai,* Ming-Hsi Chiang,* and Chien-Ming Lee*
一氧化氮 (NO) 與次硝酸 (HNO) 在生理學過程中扮演著極重要之角色,NO 與 HNO/NO- 之關鍵受體通常是由含鐵與硫配位基組成且為具生物活性之錯合物。例如,以含 NO 鍵結血質基鐵之水溶性鳥苷酸環化酶(sGC)調控內生性環鳥嘌呤單磷酸核苷(cGMP)生成是神經調節與血管舒張的關鍵步驟。除此之外,HNO 也能引發血管舒張並且比 NO 呈現更好的反應,因此可被視為治療心臟衰竭的潛在強心劑。因為 NO/HNO 在生理系統上與醫藥應用的重要性啟發,許多研究專注在金屬配位的NO或HNO釋放反應物開發上。外生性 Fe-NO 的 NO 釋放可以來自酸鹼度的改變、溶劑效應、熱或光。然而從 Fe-NO(M(HNO/NO-)) 產生之 HNO 反應仍然相對少被研究。在此著作研究中,我們開發了一特殊含硫醇之{FeNO}6 化合物 1,以可見光照射可誘發 NO/HNO 釋放。化合物 1 中的垂掛硫醇之質子與鐵中心形成 [SH•••ON-Fe] 作用可以作為 NO trap,最終在持續的激發之下而產生HNO。反應機制經由泛涵密度理論(DFT)的探究顯示,化合物 1 的可見光吸收為配位基至金屬中心的電子轉移 (LMCT) 與配位基至配位基 (NO) 的電子轉移 (LLCT),因此減弱了 Fe-NO 鍵結。重要的是,在硫醇之質子與 NO 靠近的過程中會發生電子自旋交叉 (Spin crossover),從低自旋 (Low spin) 組態轉變為自由能量較低的高自旋 (High spin) 組態。這些機制探討結論充足的支持了實驗中的觀察現象,化合物 1 在可見光的激發之下生成HNO。實驗與理論的結果顯示了分子內垂掛之質子對 HNO 生成的重要性。
We report two light-sensitive {FeNO}6 complexes, [Fe(NO)(TMSPS2)(TMSPS2H)] (1) with a pendant thiol. Complex 1 simultaneously possesses a NO group and a proton donor, which appears to be a favorable situation for HNO formation. Electronic modulation of the Fe−NO center by light irradiation for complex 1 results in the generation of NO radicals. The substituent in the secondary coordination sphere plays a significant role in dictating the fate of the photo-promoted NO. The pendant thiol of complex 1 situated above the iron-bound NO serves as a NO trap via an intramolecular [SH···ON−Fe] interaction, facilitating the release of HNO. The results reveal the important roles of the photoexcitation and the intramolecular pendant thiol proton in the HNO production. This is also a rare example of the HNO release from an electron-deficient {MNO} core. Further studies to examine the redox chemistry of complex 1 relative to the HNO production are ongoing.