Institute of Chemistry, Academia Sinica – Research
Directions
The research thrusts of the Institute are currently grouped along three major directions: materials chemistry, chemical synthesis and catalysis, and chemical biology. The current topics of materials sector include organic electroluminescent materials and devices, organic field-effect transistor materials and devices, photovoltaic materials and devices. The chemical catalysis and synthesis sector is focusing on the development of new synthetic methodology, drug discovery, carbohydrate chemistry, and the development of new catalytic systems for the generation of renewal energies and green fuels. The chemical biology program has made important advances in the delineation of bio-macromolecular structures and the development of new analytical platforms for disease detection and diagnosis.
Materials Chemistry: Organic Electronic and Optoelectronic Materialsy
Applications of organic optoelectronic materials and molecular engineering of nanomaterials are two major research directions under the materials division. Noticeable results include developments of blue fluorescent molecular materials for high performance organic light-emitting diodes, rational design of field-effective organic memory devices based on pentacene and gold nanoparticles, the very first stable organic thin film transistor based on single crystal of hexacene, rare single-walled metal–organic nanotube (MONT) with a large exterior wall diameter, the applications of metal-organic framework as optoelectronic materials, and a number of high performance materials for efficient solar energy harvesting devices such as dye-sensitized solar cells, perovskite solar cells, or organic photovoltaics. Researchers in this sector also develop stimuli-responsive materials, core-shell nanomaterials, and biomaterials. A recent report shows that a cell membrane–mimicking conducting polymer is capable to integrate biochemical and electrical stimulation to promote neural cellular behavior with great enhancement of neurite outgrowth on this conducting polymer.
Chemical Catalysis and Synthesis: Green Catalysis and Synthetic Methodology
In response to the increasingly demands of sustainable fuel and green synthetic technology, researchers in the organic synthesis and chemical catalysis divisions have strived to advance the development of cutting-edge technology for chemical transformations. The synthetic chemistry division of this sector focuses on the advances of synthetic methodology and drug discovery. The research topics under catalysis division is reconciling to catalysis relating to renewable energy. Major research directions in this sector include: (1) synthetic methodology: silyl ethers for hydroxy- directed nucleophilic acyl alkylation, microwave-assisted carbohydrate synthesis, smart fluorescent probes for bioorthogonal sugar labeling; (2) coordination chemistry: approaching unconventional catalysis via amino-NHC and carbodicarbene, unconventional porphyrin complexes for small molecule activations, engineering cytochrome P450 BM3 and alkane hydroxylase (AlkB) for alkane oxidations; (3) renewable energy catalysis: catalytic hydrogen evolution and mechanistic studies, encapsulated tricopper cluster for methane to methanol conversion, and novel catalysts for valorization of lignocellulosic biomass feedstocks.
Chemical Biology: New Material and Method towards Sustainable Health
Chemical biology division focus on the development of new material and methodology to explore the structure and function of macromolecules associated with cellular function or human diseases. The research activities are directed to unravel the underlying pathological mechanism and to derive new diagnostic and therapeutic strategies. Research topics in this division cover (1) development of smart biomaterials based on novel molecular principles; (2) chemical probe and advanced techniques in bio-imaging and structural biology; (3) drug discovery in cancer, infectious and neurodegenerative diseases; (4) development of structural biology techniques for infectious diseases, and (5) development of advanced proteomics strategies for biomarker discovery. The major achievements from the chemical biology group include the establishment of multiplexed quantitative strategy for membrane proteomics and post-translational modification for delineating disease mechanism and mining therapeutic targets discovery of amyloid fibrils induced from the TDP-43 in the Amyotrophic Lateral Sclerosis (ALS), and development of a photocontrollable probe to induce TDP-43 aggregates in live cells, mapping of the RNA exit channel on transcribing RNA polymerase II by FRET analysis, development of nano velcro chip to capture circulating tumor cells for liquid biopsy, construction of a near-infrared- activatable enzyme platform using an up-conversion nanoparticle to remotely trigger intracellular signal transduction.
負載於水滑石氧化物之原子級分散銅觸媒的合成並應用於選擇性加氫5-羥甲基糠醛生成2,5-雙(羥甲基)呋喃
Facile Synthesis of the Atomically Dispersed Hydrotalcite Oxide Supported Copper Catalysts for the Selective Hydrogenation of 5-Hydroxymethylfurfural into 2,5-Bis(hydroxymethyl)furan
Appl. Catal. B Environ. 2023, 329, 122547.
Raju Kumar, Hsin-Hui Lee, En Chen, Yuan-Peng Du, Chan-Yi Lin, Warot Prasanseang, Thanasak Solos, Kittisak Choojun, Tawan Sooknoi, Rui-Kun Xie, Jyh-Fu Lee, Po-Wen Chung*
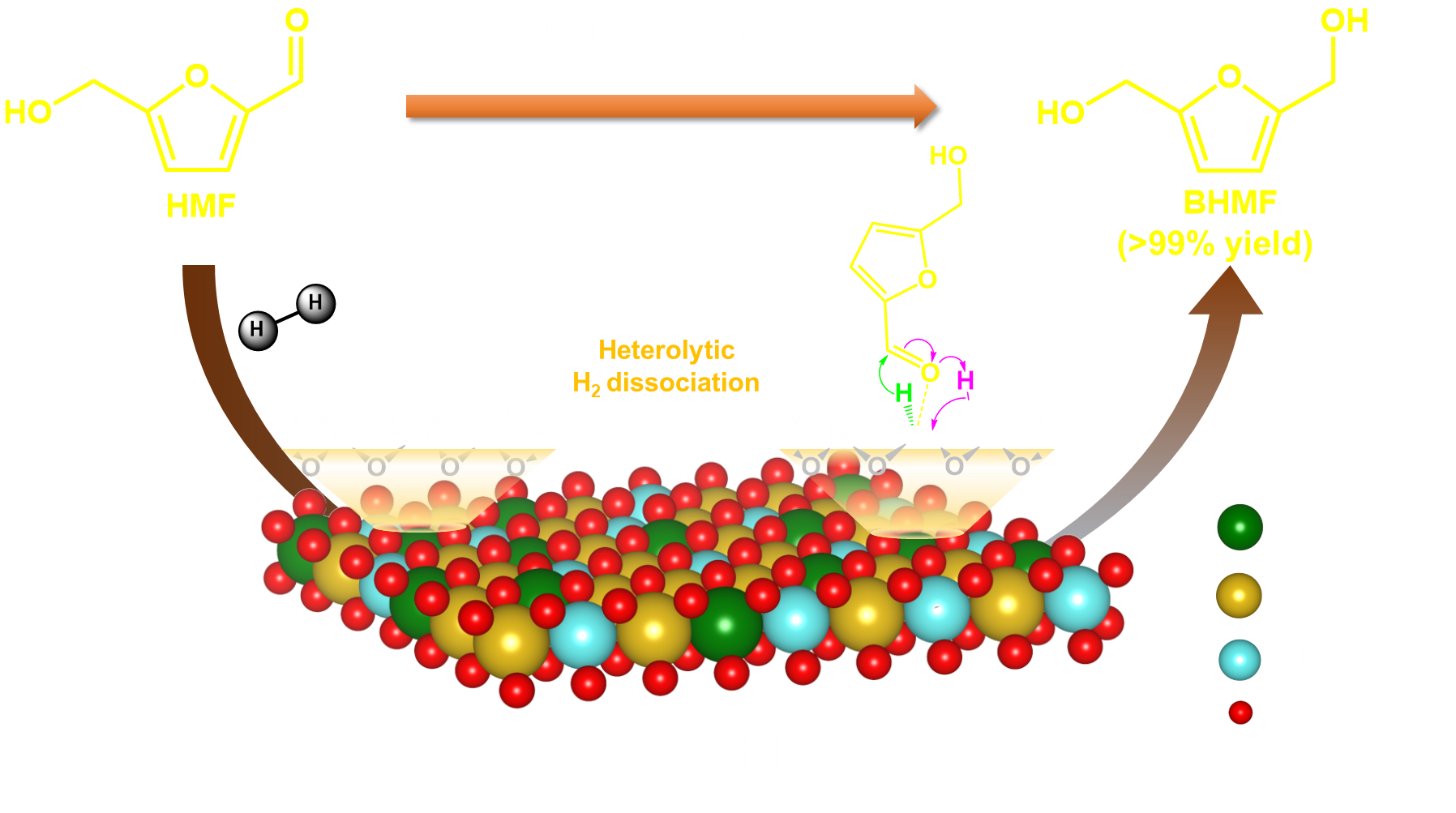
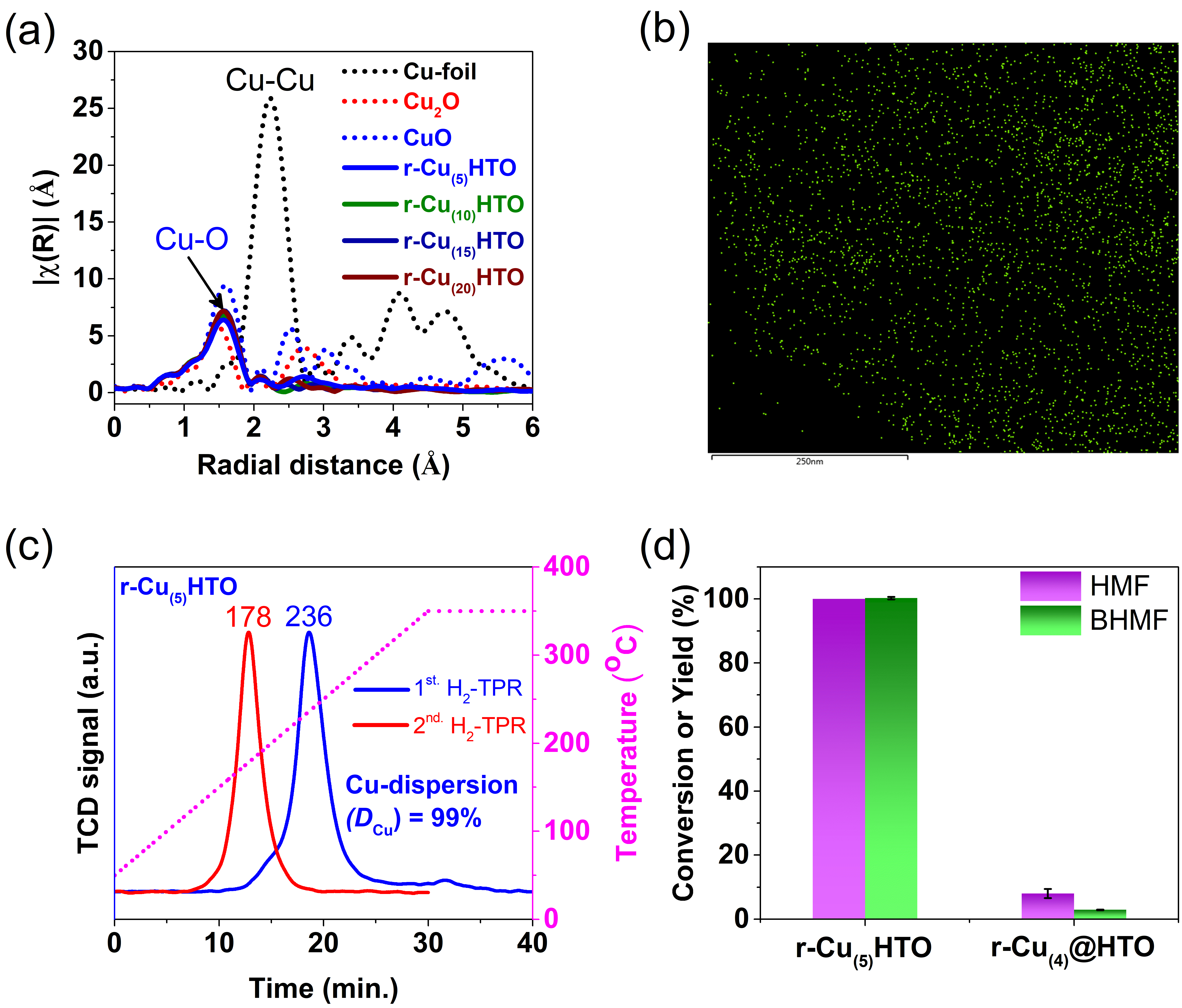
Figure 1. (a) XAS (R-space (no phase correction)) spectra of Cu-foil, CuO, Cu2O, r-Cu(x)HTO, (b) HAADF-STEM micrograph of r-Cu(5)HTO with Cu elemental mapping, (c) H2/N2O/H2-TPR chemisorption profile of r-Cu(5)HTO, and (d) HMF conversion and BHMF yield over r-Cu(5)HTO and r-Cu(4)@HTO catalysts (Reaction conditions: catalyst (10 mg) in 4 mL IPA (0.08 mmol HMF) under 5 bar of H2 at 125 °C for 1 h).
本所鍾博文副研究員團隊研發出一簡易合成技術能將銅觸媒以原子等級分散於混和氧化物的表面。我們以數種表面鑑定分析,確認了單核銅原子確實存在於水滑石氧化物載體表面,並發現在銅含量高達 20 mol% 的總金屬含量下仍可維持其原子級分散性。此銅觸媒在溫和條件下對 5-(羥甲基)糠醛的氫化反應具備高活性,並以高選擇性生成 2,5-雙(羥甲基)呋喃。其卓越的催化表現由實驗證明並歸因於觸媒表面的單核銅原子活性點,在此活性點上,異相解離的 H2 分子可對 5-(羥甲基)糠醛的羰基進行加氫反應,以達到高活性與選擇性。
Website at: https://www.sciencedirect.com/science/article/pii/S092633732300190X
The rational design of catalysts for biobased chemical production from biomass is crucial for achieving decarbonization and sustainable technologies. Recently, catalysts based on hydrotalcite-derived oxides (HTO) materials have gained attention due to their facile tailorable physicochemical properties. Atomically dispersed supported metal catalysts have emerged as intriguing materials which enable to bridge between homogeneous and heterogeneous catalysis, offering unique structural and electronic properties that enhance selective catalysis. However, these materials often require multiple synthesis steps and limited to low active metal loading. In this study, we successfully synthesized an atomically dispersed HTO supported copper catalyst (r-Cu(x)HTO; x = mol% of Cu) with up to 20 mol% copper content. The catalyst was prepared through a straightforward coprecipitation method of metal precursors in a methanolic solution at tuned pH. Comprehensive characterization techniques, including powder X-ray diffraction (PXRD), transmission electron microscopy (TEM), chemisorption (H2/N2O/H2-temperature programmed reduction (TPR)), and X-ray absorption spectroscopy (XAS), confirmed the presence of atomically dispersed copper on the HTO surface. XAS measurements specifically demonstrated the formation of mononuclear copper species in the r-Cu(x)HTO catalysts, as depicted in Figure 1a. Furthermore, H2/N2O/H2-TPR analysis, well supported by TEM (Figure 1b), revealed the dispersion of copper atoms >99% on the surface of r-Cu(5)HTO catalyst, as shown in Figure 1c. Importantly, the reduced Cu(5)HTO (r-Cu(5)HTO) catalyst exhibited excellent hydrogenation activity, completely converting HMF into BHMF under mild conditions. In contrast, the HTO supported copper nanoparticles (r-Cu(4)@HTO) displayed poor reactivity, as shown in Figure 1d. The superior catalytic performance of r-Cu(5)HTO can be attributed to the finely dispersed copper species, which selectively hydrogenate the carbonyl group (C=O) of HMF using heterolytic dissociated H2 molecules.
