Institute of Chemistry, Academia Sinica – Research
Discovery of a Dual Function Cytochrome P450 that Catalyzes Enyne Formation in Cyclohexanoid Terpenoid Biosynthesis
Angew. Chem., Int. Ed. 2020, 59, 13537–13541. Yu-Rong Chen, Annavareddi Naresh, Suh-Yuen Liang, Chun-Hung Lin, Rong-Jie Chein*, Hsiao-Ching Lin*
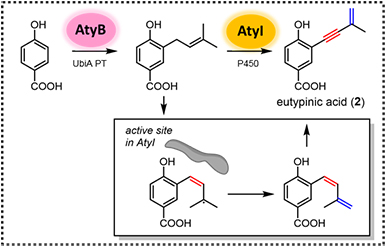
共軛烯炔是有機化學中重要的官能基之一,其在有機合成、材料和藥物等研究發展中有著廣泛的應用。目前雖有許多化學合成方法可合成共軛烯炔,但對於自然界中生物體如何催化烯炔的形成仍了解甚少。本合作研究團隊確認了在真菌 Aspergillus sp. PSU-RSPG185 的 asperpentyn 生物合成途徑,並發現了一個獨特的 cytochrome P450 酶。該酶具有雙重功能,首先催化異戊烯鏈的脫氫反應形成順式二烯中間體,接著再氧化內側雙鍵,進而生產共軛烯炔。本研究利用有機化學方法合成一系列化合物,作為起始物衍生物以及中間體類似物,進而解析此酶對於共軛烯炔的生物合成催化機制。另外,此研究也鑑定了一個非典型 UbiA 異戊烯基轉移酶,偏好轉移具五個碳的異戊烯鏈 (prenyl chain) 至對羥基苯甲酸 (p-hydroxybenzoic acid) 上。本研究不僅為真菌環己酮萜類的生物合成提供了重要的新見解,同時也提供一個新穎合成方式—以酶工具合成共軛烯炔類化合物。
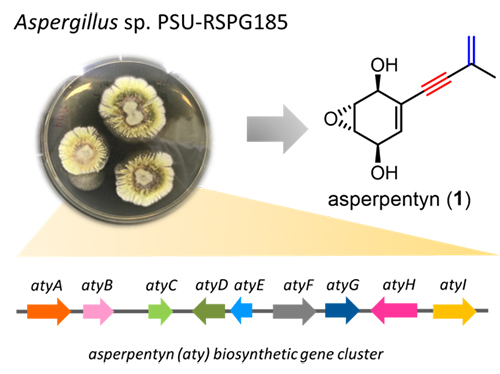
Enynes are an important functionality in organic chemistry because of their broad applications in organic synthesis, functional materials, and medicinal chemistry. Although many synthetic strategies have been developed to construct 1,3-enynes, little is known about the way nature generates this motif. In this study, we reported the identification of the asperpentyn (aty) biosynthetic gene cluster from Aspergillus sp. PSU-RSPG185. A unique P450 monooxygenase that has a dual function was discovered to first catalyze dehydrogenation of the prenyl chain to generate a cis-diene intermediate and then shows acetylenase activity to yield an alkyne moiety, giving the 1,3-enyne. The catalytic mechanism has also been characterized by using a series of substrate analogs and intermediate mimics. We also characterized an unusual UbiA prenyltransferase, that installs a short (C5) prenyl chain to a p-hydroxybenzoic acid acceptor. The studies were performed with the combination of the Saccharomyces cerevisiae heterologous expression system, in vitro enzymatic assays and synthesized substrate analogs. Collectively, these results not only provide critical new insights into the biosynthesis of fungal cyclohexanoid terpenoids, but also offer a potential route for the de novo biosynthesis of terpenoids with a 1,3-enyne functionality.