Institute of Chemistry, Academia Sinica – Research
布氏酸調控同碳雙碳烯應用於非過渡金屬協同催化聚合反應
Synergistic Catalysis by Brønsted Acid/Carbodicarbene Mimicking Frustrated Lewis Pair-Like Reactivity for Metal-free Polymerization ProcessAngew. Chem., Int. Ed. 2021
Yi-Chen Chan, Yuna Bai, Wen-Ching Chen, Hsing-Yin Chen, Chen-Yu Li, Ying-Yann Wu, Mei-Chun Tseng, Glenn P. A. Yap, Lili Zhao*, Hsuan-Ying Chen*, Tiow-Gan Ong*
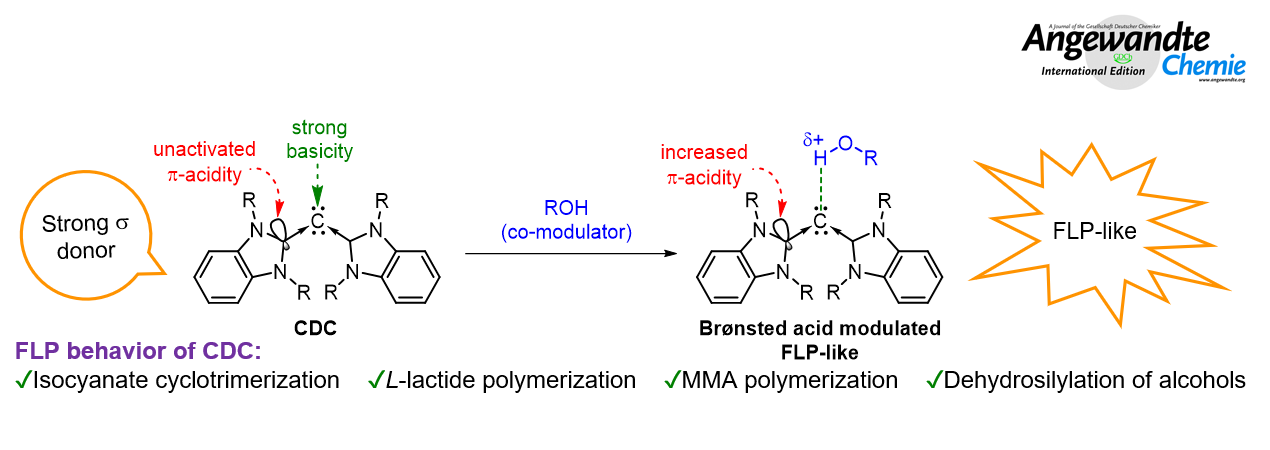
A new class of reactive non-metallic chemistry, namely Frustrated Lewis Pair (FLP) is based on a long-range attractive force between two components of Lewis acid and base that are sterically prevented from forming the classical Lewis acid-base adducts. FLP possesses unique property as it undergoes small molecule activation and metal-free catalysis like hydrogenation, CO2 reduction, hydrosilylation, borylation, amination, etc. Owing to its wide applications, FLP has become an emerging science in sustainable catalysis that reduces the dependency on fossil fuels and costly and toxic transition metal. Parallelly, carbodicarbene, a new class of carbon family, bearing two opposite reactive sites (nucleophilic and electrophilic) working simultaneously as a single component can also emulate FLP-like character for catalytic applications.
Prof. Dr. Tiow-Gan Ong at Institute of Chemistry, Academia Sinica and National Taiwan University, previously had led his research team in pioneering advanced techonology for the synthesis of wide variety of carbodicarbene. Recently, Dr. Ong research team consisting of Yi-Chen Chan, Ph.D. student and Dr. Wen-Ching Chen closely collaborated with Professor Hsuan-Ying Chen at Kaohsiung Medical University had employed alcoholic co-modulator to regulate the catalytic activity of carbodicarbene. The synergistic proof-of-concept using carbodicarbene/alcohol system successfully scores unprecedented results in dehydrosilylation, isocyanate cyclotrimerization, L-lactide (LA) and methyl methacrylate (MMA) polymerization.
The experimental efforts were also carried out to understand the enigma of these non-metallic reactions as well as the non-covalent interaction between CDC and BnOH; together with the collaborative computational analysis from Prof. Dr. Lili Zhao offers a pictorial glimpse into road-map and energy profile of this unique reaction. Such a high reactivity in this non-enzymatic catalytic system is attributed to cooperative simple hydrogen bonding and π-π interaction between carbodicarbene and alcohol derivatives.
Several aspects of this work should have broad future impacts on (i) non-metallic organic catalyst and (ii) enrichment and diversification in FLP reactions by adding different “co-modulator” molecules bearing variable functionalities. Immediate translative work is that the carbodicarbene/alcohol system will be applied into the polymerization and co-polymerization process, innovating and building new kind of recycling polymeric materials. At current stage, pilot scale polymerization is under attempt. We are looking forward to form a collaborative alliance with the chemical and material industry for metal-free polymerization in green fashion.
Yi-Chen Chan from Institute of Chemistry, Academia Sinica is the first author of the publication, working closely with team members Dr. Wen-Ching Chen, Professor Dr. Hsuan-Ying Chen, Prof. Dr. Tiow-Gan Ong (also as the principle investigator in charge of this project). The computational analysis with energy profile is solely carried out by Lili Zhao and her team members at Nanjing Tech University. The publication Synergistic Catalysis by Brønsted Acid/Carbodicarbene Mimicking Frustrated Lewis Pair-like Reactivity can be downloaded from Angewandte Chemie website as the attached link below:
https://onlinelibrary.wiley.com/doi/10.1002/anie.202107127
Media Contact:
Dr. Tiow-Gan Ong, Research Fellow/Professor, Institute of Chemistry, Academia Sinica
Email: tgong@gate.sinica.edu.tw
(Tel) +886-2-5572-8648
“受阻”路易斯酸鹼對 (frustrated Lewis pair, FLP) 是一種藉由立體障礙阻絕路易斯酸與鹼結合形成傳統酸鹼加成物的獨特結構,它能夠扮演過渡金屬的角色進行氫氣活化、二氧化碳還原、矽氫化、硼基化及胺基化等小分子活化反應。由於其廣泛的應用性,FLP已成為永續催化領域中的一門新興科學,可以減少對化石燃料及價格昂貴且具有毒性的過渡金屬的依賴。同碳雙碳烯 (carbodicarbene, CDC) 是含碳家族新一代的配位基,在其分子中具有兩個相反的反應位點 (親核性及親電性),當同時進行作用時,可以使CDC具有極為類似FLP的性質,應用於各種小分子活化及催化反應中。
中央研究院化學研究所暨台灣大學化學系的王朝諺研究員先前曾經開創了合成多樣性CDC化合物的先進技術。最近,他和所帶領的研究團隊-陳玉珍博士研究生、陳文清博士與高雄醫學大學的陳喧應教授合作,應用CDC分子結合 ”共調節劑” 苯甲醇 (布侖斯惕酸) 去調控催化活性,將這種協同效應的概念應用於醇類化合物的去氫矽基化反應 (dehydrosilylation)、異氰酸酯衍生物的環三聚合反應 (cyclotrimerization)、乳酸交酯 (L-lactide, LA) 的開環聚合反應及甲基丙烯酸甲酯 (methyl methacrylate, MMA) 的聚合反應,皆能得到前所未有的反應結果。
在研究過程中,除了利用多項實驗結果解開了CDC與苯甲醇之間的非共價交互作用,並與趙莉莉教授進行研究合作,利用理論計算得到此獨特反應的路線圖及能量分布圖,這種非酶催化反應中之所以會有如此高的反應性,主要歸因於CDC與苯甲醇之間氫鍵和 π-π 交互作用的共同影響。
這些研究成果將會對未來的研究產生以下幾種衝擊性:(1)CDC化合物作為有機(非過渡金屬)催化劑的廣泛應用性;(2)藉由添加具有可調控功能的分子來增加FLP反應性的多樣化。目前正持續將此催化劑系統應用於其他各類型的高分子聚合與共聚合反應過程,並且進行擴大量級的反應,期望在不久的未來可與高分子產業界合作,以綠色化學的概念進行不含重金屬的聚合反應。
本論文第一作者為中研院化學所博士研究生陳玉珍,研究團隊包括中研院化學所陳文清博士、通訊作者王朝諺研究員與高雄醫學大學陳喧應教授;能量分布的理論計算分析由南京工業大學趙莉莉教授及其研究團隊完成。本論文〈Synergistic Catalysis by Brønsted Acid/Carbodicarbene Mimicking Frustrated Lewis Pair-Like Reactivity〉可於Angewandte Chemie International Edition
網頁閱讀,論文全文 :
https://onlinelibrary.wiley.com/doi/10.1002/anie.202107127
新聞聯絡人:
王朝諺研究員/博士,中央研究院化學所
Email: tgong@gate.sinica.edu.tw
(Tel) +886-2-5572-8648
