Institute of Chemistry, Academia Sinica – Research
Directions
The research thrusts of the Institute are currently grouped along three major directions: materials chemistry, chemical synthesis and catalysis, and chemical biology. The current topics of materials sector include organic electroluminescent materials and devices, organic field-effect transistor materials and devices, photovoltaic materials and devices. The chemical catalysis and synthesis sector is focusing on the development of new synthetic methodology, drug discovery, carbohydrate chemistry, and the development of new catalytic systems for the generation of renewal energies and green fuels. The chemical biology program has made important advances in the delineation of bio-macromolecular structures and the development of new analytical platforms for disease detection and diagnosis.
Materials Chemistry: Organic Electronic and Optoelectronic Materialsy
Applications of organic optoelectronic materials and molecular engineering of nanomaterials are two major research directions under the materials division. Noticeable results include developments of blue fluorescent molecular materials for high performance organic light-emitting diodes, rational design of field-effective organic memory devices based on pentacene and gold nanoparticles, the very first stable organic thin film transistor based on single crystal of hexacene, rare single-walled metal–organic nanotube (MONT) with a large exterior wall diameter, the applications of metal-organic framework as optoelectronic materials, and a number of high performance materials for efficient solar energy harvesting devices such as dye-sensitized solar cells, perovskite solar cells, or organic photovoltaics. Researchers in this sector also develop stimuli-responsive materials, core-shell nanomaterials, and biomaterials. A recent report shows that a cell membrane–mimicking conducting polymer is capable to integrate biochemical and electrical stimulation to promote neural cellular behavior with great enhancement of neurite outgrowth on this conducting polymer.
Chemical Catalysis and Synthesis: Green Catalysis and Synthetic Methodology
In response to the increasingly demands of sustainable fuel and green synthetic technology, researchers in the organic synthesis and chemical catalysis divisions have strived to advance the development of cutting-edge technology for chemical transformations. The synthetic chemistry division of this sector focuses on the advances of synthetic methodology and drug discovery. The research topics under catalysis division is reconciling to catalysis relating to renewable energy. Major research directions in this sector include: (1) synthetic methodology: silyl ethers for hydroxy- directed nucleophilic acyl alkylation, microwave-assisted carbohydrate synthesis, smart fluorescent probes for bioorthogonal sugar labeling; (2) coordination chemistry: approaching unconventional catalysis via amino-NHC and carbodicarbene, unconventional porphyrin complexes for small molecule activations, engineering cytochrome P450 BM3 and alkane hydroxylase (AlkB) for alkane oxidations; (3) renewable energy catalysis: catalytic hydrogen evolution and mechanistic studies, encapsulated tricopper cluster for methane to methanol conversion, and novel catalysts for valorization of lignocellulosic biomass feedstocks.
Chemical Biology: New Material and Method towards Sustainable Health
Chemical biology division focus on the development of new material and methodology to explore the structure and function of macromolecules associated with cellular function or human diseases. The research activities are directed to unravel the underlying pathological mechanism and to derive new diagnostic and therapeutic strategies. Research topics in this division cover (1) development of smart biomaterials based on novel molecular principles; (2) chemical probe and advanced techniques in bio-imaging and structural biology; (3) drug discovery in cancer, infectious and neurodegenerative diseases; (4) development of structural biology techniques for infectious diseases, and (5) development of advanced proteomics strategies for biomarker discovery. The major achievements from the chemical biology group include the establishment of multiplexed quantitative strategy for membrane proteomics and post-translational modification for delineating disease mechanism and mining therapeutic targets discovery of amyloid fibrils induced from the TDP-43 in the Amyotrophic Lateral Sclerosis (ALS), and development of a photocontrollable probe to induce TDP-43 aggregates in live cells, mapping of the RNA exit channel on transcribing RNA polymerase II by FRET analysis, development of nano velcro chip to capture circulating tumor cells for liquid biopsy, construction of a near-infrared- activatable enzyme platform using an up-conversion nanoparticle to remotely trigger intracellular signal transduction.
借鏡非洲爪蛙!中研院團隊解開微脂體與胜肽互動之謎,打造精準投藥「智慧型微脂體」
Inspired from the African Clawed Frog! Academia Sinica Team Unravels the Mystery of Liposome-Peptide Interactions to Create "Smart Liposomes" for Precision Drug DeliveryJ. Am. Chem. Soc. 2026, 148, 8, 8749–8760
Hua-De Gao, Jia-Lin Hong, Cheng-Bang Jian, Tzu-Ho Chen, Ning-Chu Chang, Suthasinee Meeroekyai, Ruei-Yu He, Yi-Ting Liao, Chun-Hsiung Wang, Chun-Jen Su, U-Ser Jeng, Meng-Chiao Ho, Yu-Ju Chen, Hsien-Ming Lee*
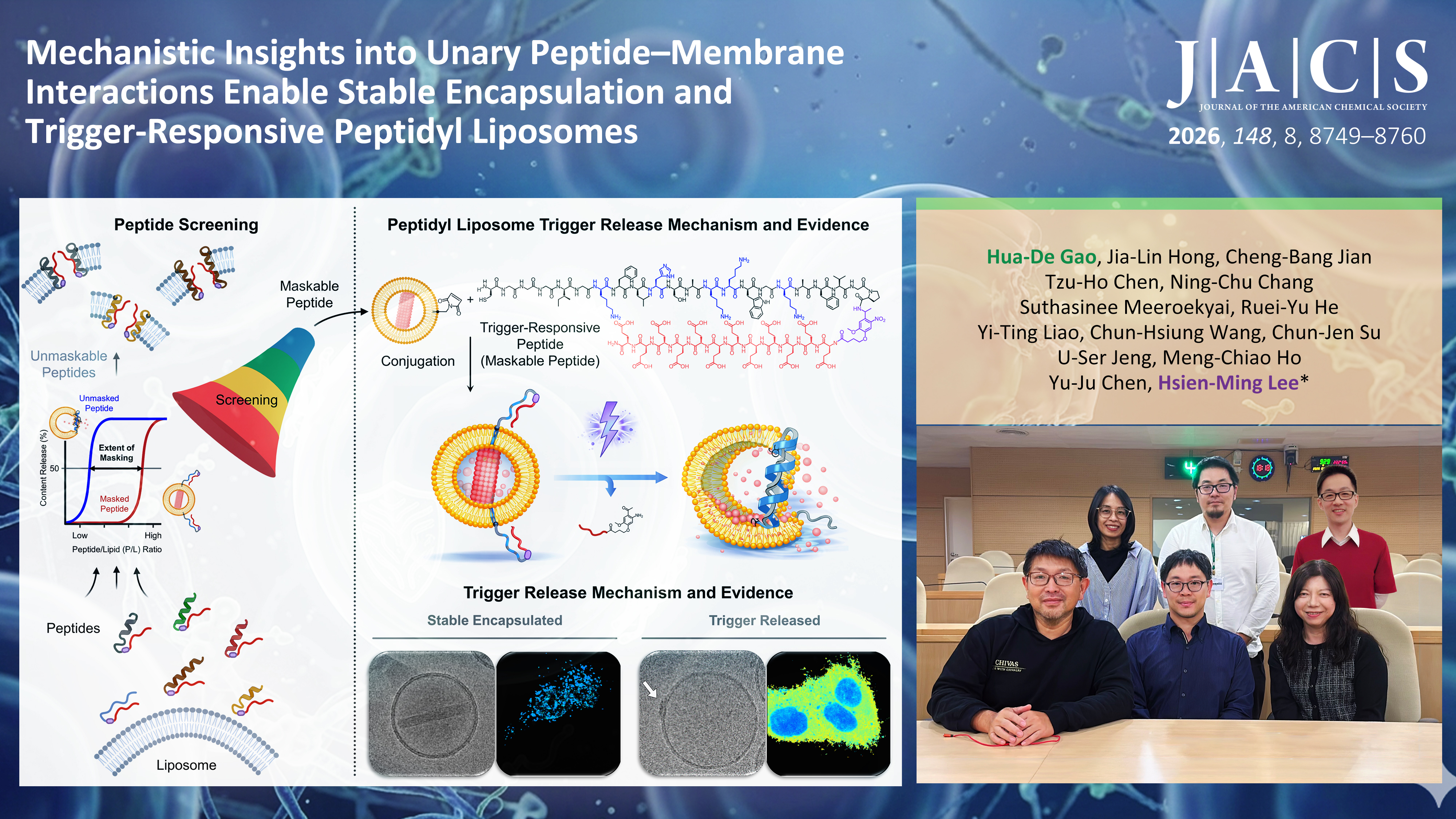
利用微脂體作為傳遞藥物載體已成為現代醫學的重要技術。將「胜肽」精準裝配於微脂體,使其如引信般作為訊號感測器,可實現多元及智慧型的精準釋放。然而,胜肽與微脂體膜結合後,其分子交互作用非常複雜,相關機制仍有許多未知。在過去研究中,科學家多半將胜肽與微脂體分開研究,這稱為「二元系統」。可是,一旦將胜肽直接連接到微脂體表面(即「一元系統」),多肽修飾的微脂體往往會變得極不穩定,導致藥物提前釋放,此一穩定性問題一直是難以克服的重大瓶頸。
為了突破此困境,中央研究院化學所李賢明副研究員帶領的研究團隊,系統性篩選多種具破膜能力的胜肽,希望能尋找一種在平時可維持微脂體穩定、接收訊號能精準啟動的理想多肽骨架,同時釐清其與微脂體膜的關鍵互動機制。研究團隊發現,一種源自「非洲爪蛙」表皮分泌的抗菌胜肽,正是突破的關鍵。在二元系統中,此胜肽對雙電性微脂體幾乎不具破壞性;在一元系統中,它卻展現出強大的功能性,這是以前所未知的。透過特殊的化學屏蔽設計,該胜肽是目前唯一可穩定停留於微脂體表面不引發洩漏,但在接收訊號後能成功啟動破膜活性的胜肽骨架,成功兼顧系統穩定性與釋放效率。
透過冷凍電子顯微鏡、小角X光散射及螢光顯微成像等多種尖端技術,研究團隊清晰捕捉了這個微觀世界的動態過程。當智慧微脂體接收到特定訊號時,表面的胜肽隨即被喚醒,在膜表面移動並逐步聚集,進而於局部精準形成暫時性缺口,使藥物得以順利釋放。此發現重新定義了引信式釋放多肽微脂體的設計,未來可望提升臨床精準投藥的安全性與效率。
本研究第一作者為高華德博士(現為中研院化學所博士後研究人員,原為臺灣大學化學系之博士生)。研究結合了跨領域技術:包括冷凍電子顯微術(中研院生化所何孟樵副研究員)、小角 X 光散射(國家同步輻射研究中心鄭有舜教授及蘇群仁副研究員) 、及中研院化學所質譜與影像核心設施及冷凍電子顯微鏡設施之技術協助。本研究感謝中研院材料與分析科技探索計畫(iMATE)與國家科學及技術委員會(NSTC)的經費支持。研究成果已於 2026 年 2 月 18 日發表於《美國化學會誌》(Journal of the American Chemical Society)。
Utilizing liposomes for drug delivery is a vital technology in modern nanomedicine. Equipping the liposome surface with "peptides" to act as signal transducers works much like a trigger, enabling diverse and smart precision-release designs. However, the molecular interactions between peptides and liposomal membranes following chemical conjugation are highly complex, with related mechanisms remaining largely unknown. In past studies, scientists mostly studied peptide-liposome interactions as two chemical entities, which is known as a "binary system". Yet, once a membrane-active peptide is directly conjugated to the liposome surface (forming a "unary system"), peptide-modified liposomes often become highly unstable, leading to premature drug leakage—this encapsulation stability issue has been a major bottleneck that is difficult to overcome.
To break through this dilemma, the research team led by Associate Research Fellow Hsien-Ming Lee at the Institute of Chemistry, Academia Sinica, systematically screened various membrane-lytic peptides. The goal was to find an ideal peptide backbone that maintains liposome stable in the signal waiting state and activates precisely upon receiving a signal, while simultaneously elucidating its key interaction mechanisms with the liposome membrane. The research team discovered that an antimicrobial peptide (AMP) secreted from the skin of the "African clawed frog" was the key to this breakthrough. In binary systems, this peptide exhibits almost no destructive power against liposomes; however, in a unary system, it demonstrates powerful functionality. Through a specialized chemical masking design, it is currently the only AMP backbone capable of remaining fully inert when masked on the surface without inducing leakage, while activating its membrane-lytic activity upon receiving a signal, successfully balancing liposome encapsulating stability and release efficiency.
By employing cutting-edge techniques such as cryo-electron microscopy, small-angle X-ray scattering, and fluorescence-lifetime imaging microscopy, the research team clearly captured the dynamic processes of this microscopic world. When this smart peptidyl liposome receives a triggering signal, the surface peptides are immediately awakened, moving and gradually aggregating on the membrane surface, and then creating a membrane defect to release the drug. This discovery redefines the design principles of trigger-responsive peptidyl liposomes, and is expected to significantly enhance the feasibility of clinical peptide-modified liposome for precision drug delivery in the future.
The first author of this study is Dr. Hua-De Gao (currently a postdoctoral researcher at the Institute of Chemistry, Academia Sinica, and originally a Ph.D. student at the Department of Chemistry, National Taiwan University). The research integrated cross-disciplinary technologies, including cryo-electron tomography (Prof. Meng-Chiao Ho, Institute of Biological Chemistry, Academia Sinica), small-angle X-ray scattering (Prof. U-Ser Jeng and Prof. Chun-Jen Su, National Synchrotron Radiation Research Center), and technical assistance from the Mass Spectrometry and Imaging core facilities at the Institute of Chemistry and the Academia Sinica Cryo-EM Facility. This study gratefully acknowledges financial support from the Academia Sinica iMATE Program and the National Science and Technology Council (NSTC). The research findings were published in the Journal of the American Chemical Society on February 18, 2026.
